The Ultimate Guide to Corrosion Protection: Pickling, Plating, and Galvanizing
Pickling, plating, and galvanizing are three common and widely used methods to protect metal from corrosion. Pickling is a critical preparatory step that removes surface impurities to ensure strong adhesion for subsequent coatings, with no standalone corrosion protection effect. Plating deposits a thin, precise metal layer via electrochemical means, delivering corrosion resistance plus enhanced properties like hardness and aesthetics, suitable for mild, controlled indoor environments. Galvanizing bonds a thick sacrificial zinc layer through metallurgical or electrochemical reactions, offering long-term protection for steel and iron in harsh outdoor/industrial settings.
This ultimate guide explains these three methods in detail, including clear explanations of how each works, their key properties and performance, cost considerations, and real-world applications.
What Are Pickling, Plating, and Galvanizing?
Pickling, plating, and galvanizing are surface treatment processes. They modify or coat metal to create a barrier against corrosive forces.
1. Pickling
Pickling is used to remove oxide scales, rust, mill scale, grease, and other surface impurities from ferrous metals such as steel and iron, as well as non‑ferrous metals including aluminum and copper. It is mainly a preparatory process: a clean metal surface is necessary for strong bonding with later coatings like plating or galvanizing. Without pickling, coatings may peel, bubble, or fail to provide full corrosion protection.
The pickling process uses acidic solutions called “pickling liquor,” which react with and dissolve surface oxides and contaminants without harming the base metal. The type of acid used depends on the kind of metal being treated.
- Hydrochloric acid (HCl): Use for carbon steel, stainless steel and iron.
- Property: Fast-acting, low-cost, easy to neutralize.
- Importance: Fast reaction boosts efficiency, easy neutralization avoids residual acid rust, low cost fits mass production—its core properties together make it the most widely used pickling acid.
- Sulfuric acid (H₂SO₄): Use for steel with thick oxide scales and aluminum pickling.
- Property: Strong scale-dissolving ability, prone to over-etching non-ferrous metals.
- Importance: Strong dissolving power targets stubborn thick oxides; over-etching risk means additives are a must for aluminum to protect the base metal.
- Nitric acid (HNO₃): Use for stainless steel, copper and brass.
- Property: Creates smooth, passivated surfaces.
- Importance: Smooth finish meets precision part requirements; passivation forms a basic anti-corrosion layer, laying a good foundation for subsequent coatings.
- Phosphoric acid (H₃PO₄): Use for light rust removal and prepping metals for painting/plating.
- Property: Mild, low-etching, forms a bonding layer on metal surfaces.
- Importance: Mildness prevents base metal damage; the bonding layer it forms greatly improves the adhesion of subsequent paint or plating layers.
After the acid treatment, the metal is thoroughly rinsed with water to remove all acid residue, then dried and often neutralized with a basic solution to prevent re‑rusting. The result is a clean, smooth, and chemically active surface that is ready for further corrosion protection or fabrication.
Pickling can be done in batch tanks for small or irregularly shaped parts, or in continuous lines for large sheets, bars, or pipes in high‑volume production. It is a post-manufacturing or pre-coating step. It does not provide long‑term corrosion protection by itself, but it is a foundational step that makes other protective methods work effectively.
2. Plating
Plating, also called electroplating, is an electrochemical process that deposits a thin, uniform layer of corrosion-resistant metal onto a base metal part.The plated layer acts as a physical or sacrificial barrier, depending on the metal used, protecting the base metal from corrosive elements. It also improves other properties such as hardness, wear resistance, conductivity, and appearance.
The electroplating process takes place in an electrolytic bath and involves three key components:
- The anode: A piece of the plating metal (e.g., nickel, chrome, copper, zinc) that dissolves into the bath, releasing metal ions.
- The cathode: The base metal part to be plated, which is connected to the negative terminal of a power supply.
- The electrolytic solution: A liquid containing dissolved salts of the plating metal, which conducts electricity and allows metal ions to travel from the anode to the cathode.
When an electric current is applied, metal ions from the anode are attracted to the negatively charged cathode and form a thin, adherent layer on the base metal surface.
The thickness of the plated layer is controlled by current duration and voltage. Typical thickness ranges from a few microns for decorative parts to over 50 microns for industrial components that need strong corrosion protection.
The most common plating metals used for corrosion protection are:
- Zinc: The most widely used plating metal for steel and iron, affordable and effective for general corrosion protection in mild environments (e.g., indoor equipment, automotive fasteners).
- Nickel: Provides excellent corrosion resistance and a smooth, bright finish; often used as a base layer for chrome plating or on its own for food processing, medical, and aerospace parts.
- Chrome (chromium): Known for its hard, scratch-resistant, and corrosion-resistant surface; used for automotive parts (bumpers, wheels), hydraulic cylinders, and industrial machinery.
- Copper: A conductive, malleable plating metal used as a base layer for nickel/chrome plating (to fill in surface imperfections) and for corrosion protection of electrical components.
- Tin: A food-safe plating metal used for steel cans and food processing equipment, as it is non-toxic and resistant to organic acids.
- Gold/Silver: Precious metals used for high-end corrosion protection and conductivity in electronics, aerospace, and medical devices (cost-prohibitive for general industrial use).
Plating can also be done through electroless plating, a chemical process that does not require electricity.
It is suitable for complex, irregularly shaped parts such as gears and fasteners, where electroplating may result in uneven coverage.
Electroless nickel plating is widely used for industrial components, as it forms a uniform layer even on blind holes and intricate surfaces.
3. Galvanizing
Galvanizing is a hot-dip coating process that applies a thick, durable layer of zinc or zinc-alloy to steel or iron, providing long-term sacrificial corrosion protection.
Unlike electroplating, which deposits a thin layer electrochemically, galvanizing bonds a thick zinc layer to the metal surface through a metallurgical reaction.This forms a zinc-iron alloy layer at the interface, ensuring strong adhesion and high durability.
The hot-dip galvanizing process follows a strict sequence of steps (pickling is always a critical pre-step here):
- Surface preparation: The steel/iron part is cleaned via pickling (to remove oxide scale and rust) and degreasing (to remove oil/grease), then rinsed and dried.
- Fluxing: The clean metal is dipped in a zinc chloride flux solution to prevent re-oxidation and ensure the molten zinc wets the metal surface evenly.
- Hot-dip coating: The metal is immersed in a bath of molten zinc (at approximately 450°C/842°F) for a few minutes. The zinc reacts with the iron in the steel to form a series of zinc-iron alloy layers (the intermetallic layer) on the surface, with a pure zinc layer on top.
- Cooling and finishing: The part is removed from the zinc bath, drained of excess zinc, and air-cooled (or quenched in water). The finished surface is a matte, spangled zinc layer that can be left as-is or painted/coated for additional protection or aesthetics.
A newer variation is electrogalvanizing, which uses an electrochemical process similar to plating to deposit a thin zinc layer onto steel. It creates a smoother, more uniform surface than hot‑dip galvanizing, making it ideal for parts that need painting or precise dimensional tolerances. Hot‑dip galvanizing, by contrast, provides a thicker, more durable layer suitable for harsh outdoor environments.
Zinc is the main metal used in galvanizing because it is sacrificial. When exposed to moisture and oxygen, zinc corrodes before the underlying steel or iron. This sacrificial protection continues to guard the base metal even if the zinc layer is scratched or damaged, giving galvanizing a key advantage over many plating methods. Zinc also forms a tough, insoluble layer of zinc oxide and zinc hydroxide that acts as a physical barrier and slows further corrosion.
Galvanizing can also use zinc alloys such as zinc‑aluminum and zinc‑magnesium for better performance. Zinc‑aluminum alloys improve corrosion resistance in saltwater environments, while zinc‑magnesium alloys offer stronger adhesion for paint and other coatings.
Key Differences Between Pickling, Plating, and Galvanizing
Pickling, plating, and galvanizing differ fundamentally. They differ in purpose, process, protective performance and cost. The table below breaks down their core differences across the most important factors for project selection:
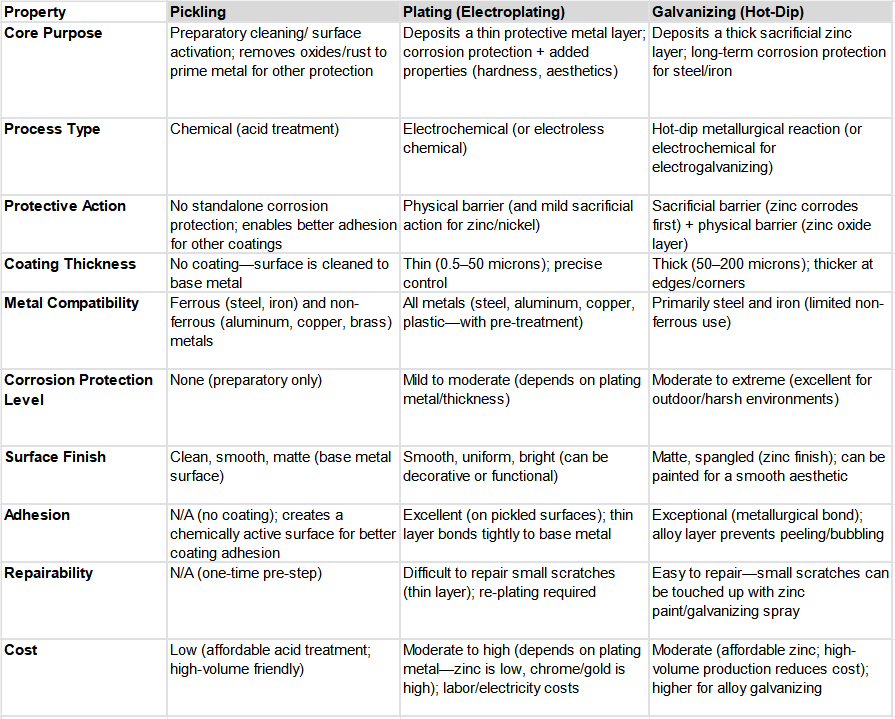
Additional Key Distinctions
- Standalone vs. Preparatory: Pickling is never used alone for corrosion protection. It is always a precursor to plating, galvanizing, painting, or fabrication. Plating and galvanizing are standalone protective methods. However, they nearly always require picking first.
- Sacrificial vs. Non-Sacrificial: Galvanizing is a sacrificial protection method. Zinc corrodes to save the base metal. Most plating methods are non-sacrificial. The plated layer acts only as a physical barrier. If scratched, the base metal corrodes. Zinc plating offers mild sacrificial protection. However, it is less effective than galvanizing due to its thinner layer.
- Dimensional Tolerances: Plating deposits a very thin layer. This makes it ideal for precision parts. Examples include gears and fasteners. These require tight dimensional tolerances. Galvanizing’s thicker layer can alter dimensions. Therefore, it is not recommended for high-precision parts.
- Environment Suitability: Plating is best for mild, controlled environments. These are indoor, low-moisture spaces. Galvanizing excels in harsh, outdoor environments. These have exposure to rain, snow, salt, and industrial chemicals.
How to Choose Between Pickling, Plating, and Galvanizing
Pickling is not a “choice” in the same way as plating and galvanizing—it is a required step for nearly all metal coating processes. The real decision is between plating and galvanizing, and this choice depends on your project’s core requirements: the metal type, service environment, corrosion risk, dimensional tolerances, aesthetic needs, and budget. Use the following framework to select the right corrosion protection solution for your application:
1. Consider the Metal Type
- Steel/Iron: The primary candidates for both plating and galvanizing. Galvanizing is the go-to for steel/iron in harsh environments, while plating (zinc, nickel) is better for precision steel/iron parts in mild environments.
- Aluminum/Copper/Brass (Non-Ferrous Metals): Only plating is a viable option—galvanizing is not compatible with non-ferrous metals, as the metallurgical reaction between zinc and these metals does not occur. Pickling is still used to prepare non-ferrous metals for plating (with metal-specific acids).
- Plastics/Composite Materials: Can be plated (with pre-treatment to make the surface conductive) but cannot be galvanized or pickled (acid treatment would damage the material).
2. Consider the Service Environment
The corrosiveness of the environment is the single most important factor. It determines the choice between plating and galvanizing:
- Harsh Outdoor/Industrial Environments: These include seawater, coastal salt air, rain and snow, industrial chemicals, and road salt. Choose hot-dip galvanizing. Its thick sacrificial zinc layer provides long-term protection (20–50+ years) in these conditions, even with minor surface damage. Zinc-aluminum alloy galvanizing is ideal for marine applications.
- Mild Controlled Environments: These include indoor equipment and low-moisture spaces. These are dry industrial settings. Choose plating. Zinc plating is the most cost-effective option for steel/iron parts, while nickel plating is better for parts requiring enhanced wear resistance or a bright finish.
- Food/Medical Environments: Choose tin or nickel plating. These are food-safe and non-toxic. Electropolished stainless steel is also an option. It is often pre-treated with pickling. Galvanizing is not recommended here, as zinc can leach into food/medical products.
- Electronics/Aerospace: Choose gold, silver, or nickel plating. These provide excellent conductivity and corrosion protection in high-precision applications. Galvanizing is too thick and heavy for these components.
3. Consider the Dimensional Tolerances and Part Geometry
- High-Precision Parts: These include gears, fasteners, bearings, and electronic components. Choose plating. Its thin, uniform layer (micron-level thickness) does not alter the part’s dimensions, ensuring a perfect fit and function. Electroless plating is ideal for complex, irregularly shaped parts with blind holes or intricate details.
- Large/Structural Parts: These include beams, pipes, tanks, automotive frames, and agricultural equipment. Choose hot-dip galvanizing. These parts do not require tight dimensional tolerances, and galvanizing’s thick layer provides durable protection for large surface areas. Galvanizing is also cost-effective for high-volume production of large parts.
- Parts Requiring Painting/Additional Coating: Choose electrogalvanizing or zinc plating. These provide a smooth surface for paint adhesion. Hot-dip galvanizing is also an option. It requires a phosphating pre-step for better paint bonding. Nickel/chrome plating is not ideal for painting, as the smooth surface resists paint adhesion.
4. Consider the Aesthetic and Functional Needs
- Decorative Parts: These include automotive trim, consumer goods, jewelry, and furniture. Choose plating. Options include chrome, nickel, copper, or gold and silver. Plating offers a smooth, bright, and customizable finish (matte, polished, brushed) that is far more aesthetically pleasing than galvanizing’s spangled zinc finish.
- Functional/Industrial Parts:These have no aesthetic requirements. Choose hot-dip galvanizing for harsh environments. Choose zinc plating for mild environments. Both are cost-effective and prioritize performance over appearance.
- Parts Requiring Additional Properties: These include wear resistance, conductivity, and hardness. Choose plating. Chrome plating offers exceptional wear resistance, copper/gold plating provides high conductivity, and nickel plating boosts hardness—galvanizing only provides corrosion protection with no added functional properties.
5. Consider the Budget Constraints
- Tight Budget + General Corrosion Protection: Choose zinc plating for mild environments. Choose hot-dip galvanizing for harsh environments. Both are the most affordable corrosion protection options for steel/iron.
- Moderate Budget + Enhanced Performance/Aesthetics: Choose nickel or electroless nickel plating. These offer better corrosion resistance and wear resistance than zinc plating, with a smooth finish.
- High Budget + High-End Applications: Choose chrome, gold, or silver plating. These are for electronics, aerospace, and medical applications. Choose zinc-alloy galvanizing for marine and extreme industrial environments. These options provide the highest level of performance but come with higher material and labor costs.
- Remember Pickling Costs: Pickling is a low-cost step. However, it is non-negotiable for plating and galvanizing. Skipping pickling will lead to coating failure and higher long-term costs for repairs/replacement.
Typical Application Scenarios
Pickling Applications (Preparatory Step)
Pickling is used in all industries that process metal and apply protective coatings. Its most common applications include:
- Preparing steel/iron sheets, bars, and pipes for galvanizing in construction and infrastructure.
- Cleaning automotive parts (frames, fasteners, body panels) for zinc/nickel plating or painting.
- Removing oxide scale from stainless steel parts in food processing and aerospace manufacturing before plating or passivation.
- Preparing non-ferrous metals (aluminum, copper) for plating in electronics and consumer goods production.
- Cleaning industrial machinery parts for electroless nickel plating to improve wear resistance.
Plating Applications
Plating is used across industries for both corrosion protection and enhanced performance/aesthetics, with the most common use cases:
- Automotive: Zinc plating for fasteners. Nickel and chrome plating for bumpers, wheels, and trim. Copper plating as a base layer for body panels
- Electronics/Aerospace: Gold and silver plating for circuit boards and connectors. This provides conductivity and corrosion protection. Nickel plating for aerospace components. This provides hardness and corrosion resistance.
- Food Processing/Medical: Tin plating for steel cans and food equipment. This is food-safe. Nickel plating for surgical tools and medical devices. This is hygienic and corrosion-resistant.
- Consumer Goods: Chrome and nickel plating for furniture hardware, door handles, and kitchen appliances. This provides aesthetics and corrosion protection. Copper plating for jewelry and decorative items.
- Industrial Manufacturing: Electroless nickel plating for gears, bearings, and hydraulic cylinders. This provides wear resistance and corrosion protection. Zinc plating for small machine parts. This provides general corrosion protection.
Galvanizing Applications
Galvanizing is almost exclusively used for steel and iron. It is used in harsh outdoor and industrial environments. Core applications include
- Construction/Infrastructure: Hot-dip galvanizing for structural beams, bridge components, streetlights, guardrails, and water/sewage pipes—provides long-term protection against rain, snow, and road salt.
- Marine/Coastal: Zinc-aluminum alloy galvanizing for boat hulls, dock pilings, and offshore platform parts—resists saltwater corrosion better than standard zinc galvanizing.
- Agriculture/Heavy Machinery: Hot-dip galvanizing for tractors, farm equipment, and industrial machinery frames—protects against mud, chemicals, and outdoor exposure.
- Automotive: Electrogalvanizing for steel body panels and frames—provides a smooth surface for painting and mild corrosion protection against road salt and moisture.
- Storage/Transport: Galvanizing for steel tanks (water, fuel, chemicals), shipping containers, and truck beds—protects against corrosion from stored liquids and outdoor transport.
FAQs
1. Which is more corrosion-resistant: plating or galvanizing?
Hot-dip galvanizing is more corrosion-resistant than most plating methods. This is especially true in harsh outdoor and industrial environments. Its thick sacrificial zinc layer (50-200 microns) provides long-term protection. This works even with minor scratches. Plating’s thin layer (0.5-50 microns) acts only as a physical barrier. The exception is zinc plating. It offers mild sacrificial protection. However, it is less effective than galvanizing. For mild, controlled environments, nickel or zinc plating provides sufficient corrosion protection at a lower cost.
2. Can galvanized metal be painted?
Yes. Galvanized metal can be painted. This provides additional corrosion protection or aesthetic appeal. However, the surface must be properly prepared. This includes degreasing, abrading, or phosphating. This ensures paint adhesion. Electrogalvanized metal has a smoother surface. It is easier to paint than hot-dip galvanized metal. Hot-dip galvanized metal has a spangled finish. Painted galvanized metal offers a dual barrier of protection. This includes the sacrificial zinc layer and the paint layer.
3. What is the lifespan of galvanized vs. plated metal?
- Hot-dip galvanized steel and iron: This has a lifespan of 20-50+ years in outdoor and harsh environments. For example, rural areas: 50+ years. Coastal salt air: 20-30 years. Industrial areas: 15-25 years.
- Zinc-plated steel and iron: This has a lifespan of 5-15 years in mild, indoor environments. The lifespan drops to 1-3 years in harsh outdoor and coastal environments. This is due to the thin zinc layer.
- Nickel and chrome-plated metal: This has a lifespan of 10-20 years in mild and moderate environments. It is excellent for indoor use. However, chrome plating can pit in saltwater and harsh chemicals.
Lifespan also depends on maintenance: regular cleaning and touch-ups for scratches will extend the life of both galvanized and plated metal.
4. What is the difference between hot-dip galvanizing and electrogalvanizing?
- Hot-dip galvanizing: This is a hot-dip process. It deposits a thick (50-200 microns) zinc layer via a metallurgical reaction. It provides excellent corrosion resistance for harsh environments. It has a matte spangled finish. It slightly alters dimensions.
- Electrogalvanizing: This is an electrochemical process. It is similar to plating. It deposits a thin (5-20 microns) zinc layer. It has a smooth, uniform finish. It has precise dimensional tolerances. It is ideal for painting. It provides moderate corrosion resistance for mild environments.
5. Does plating/galvanizing affect the weldability of metal?
- Plating: Most plating metals (zinc, nickel, copper) do not significantly affect weldability, but the plating layer may produce minor fumes during welding—ventilation is recommended. Zinc plating can cause “zinc embrittlement” in high-strength steel if welded at high temperatures, so the plating layer is often removed from weld areas.
- Galvanizing: The thick zinc layer can produce zinc fumes during welding. These are hazardous if inhaled. It can also cause porosity in the weld. Galvanized metal can be welded. However, the zinc layer is typically ground off from the weld zone first. The weld is touched up with zinc paint or galvanizing spray after welding.
Learn More About Our Products?
Contact Now


